SF6 gas is an inert, nontoxic, colorless, odorless, tasteless, and nonflammable gas. In this gas molecule six tightly bonded fluorine atoms surrounds one sulfur atom.
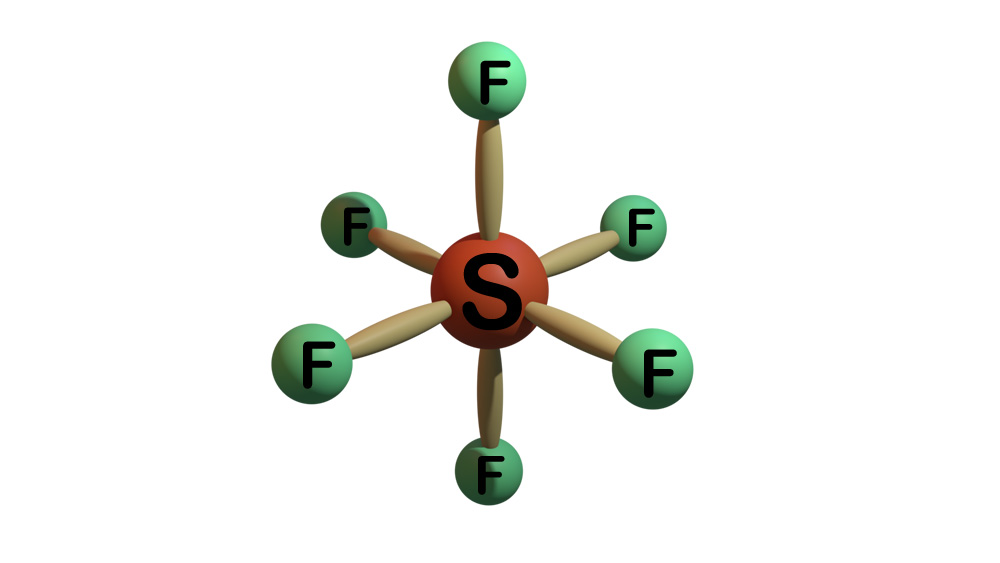
This molecular structure makes SF6 gas five times heavier than air. SF6 in GIS stays at 400 to 600 kPa of absolute pressures. This gas offers three times more dielectric strength than that of air at the same pressure. SF6 extinguishes arc 100 times better than air. It serves as the best medium for ultra high voltage interrupting medium till date. SF6 gas decomposes instantly in the high temperature of an electric arc. Although, the decomposed gas instantly recombines back into SF6.
